Design
This study employed a two-arm, single-blind randomized controlled trial (RCT) design. The trial was conducted in accordance with the CONSORT guidelines and was prospectively registered with the Chinese Clinical Trial Registry (Registration No.: ChiCTR2300075082; Date: 24/08/2023). Ethical approval was obtained from the Ethics Committee of the Affiliated People’s Hospital of Jiangsu University (Approval No. K-2023007-Y). All procedures adhered to the Declaration of Helsinki.
Participants were randomly assigned to either the intervention group (CDIP) or the control group (routine prenatal care). Outcome assessments were conducted at three time points: t1 (baseline, 12 weeks’ gestation), t2 (post-intervention, 24 weeks’ gestation), andt3 (follow-up, before delivery at 37–40 weeks’ gestation).
Instrument with validity and reliability
Nutrition Literacy was assessed using the Nutrition Literacy Assessment Instrument for Pregnant Women (NLAI-P), which demonstrated high reliability (Cronbach’s α = 0.82) and strong content validity (CVI = 0.98, CVR = 0.97). Confirmatory factor analysis supported structural validity (χ²/df = 1.82, GFI = 0.86, RMSEA = 0.046)5.
Eating Behavior was measured using the validated Chinese version of the Dutch Eating Behavior Questionnaire (DEBQ-C), with strong internal consistency (total α = 0.94; emotional α = 0.96; external α = 0.88; restrained α = 0.91)32,33.
Dietary Quality was assessed through a pregnancy-specific Food Frequency Questionnaire (FFQ-P) and evaluated using the Diet Balance Index for Pregnancy (DBI-P)34. Reliability indicators included test-retest ICCs for nutrients (0.24–0.58) and Pearson correlations for food and nutrient items (0.28–0.59)35.
GWG was tracked using standardized procedures from pre-pregnancy to delivery.
Gestational diabetes mellitus (GDM) was diagnosed based on the 75 g 2-hour oral glucose tolerance test (OGTT), conducted between 24 and 28 gestational weeks, following the criteria recommended by the Chinese Guidelines for the Diagnosis and Treatment of Pregnancy Complications (2020 edition).
Sampling and recruitment
Participants were recruited from the obstetric outpatient clinic of Changzhou No. 2 People’s Hospital between August and November 2023. A random sampling strategy was applied through the hospital’s intelligent medical system. All participants provided written informed consent before enrollment.
Inclusion and exclusion criteria
Inclusion criteria were age between 18 and 35 years, pre-pregnancy Body Mass Index (BMI) between 18.5 kg/m² and 24 kg/m², primigravida with a singleton pregnancy, gestational age no more than 12 weeks, ability to use WeChat, and at least one family member other than the pregnant woman responsible for cooking. Exclusion criteria included diabetes, uncontrolled hypertension, thyroid disorders, cardiovascular diseases, cancer, pulmonary diseases, severe gastrointestinal diseases, a history of eating disorders or bariatric surgery, severe mental illnesses, a history of mood or anxiety disorders in the past three months, substance abuse, and threatened miscarriage.
Sample size and power
Sample size was calculated using G*Power based on the expected effect size (Cohen’s d = 0.63) from a prior study36. A total of 80 participants (40 per group) were required to achieve 80% power at a 5% significance level. Accounting for a 10% attrition rate, the final sample size was increased to 88 (44 per group).
Randomization and blinding
Participants were randomly assigned to either the intervention group or the control group using simple randomization with a 1:1 allocation ratio. The random sequence was generated by a computer and implemented by an independent researcher who was not involved in participant recruitment or outcome assessment.
The intervention group received both offline and online support from trained midwives, including individualized dietary guidance and follow-up. The control group received routine prenatal care as per standard practice. To prevent contamination between groups, participants in the intervention group were physically separated during face-to-face consultations and managed independently online.
Intervention overview
Routine care
All participants received routine prenatal care in accordance with national Chinese medical guidelines, encompassing four scheduled antenatal visits between 12 and 24 weeks of gestation. These visits included standard physical examinations, biochemical assessments, and recommended screening tests.
Under routine care, pregnancy-related health education primarily addressed topics such as childbirth preparation and breastfeeding. Structured nutritional education was not routinely provided, thereby requiring pregnant women to seek dietary guidance independently from healthcare providers or external resources.
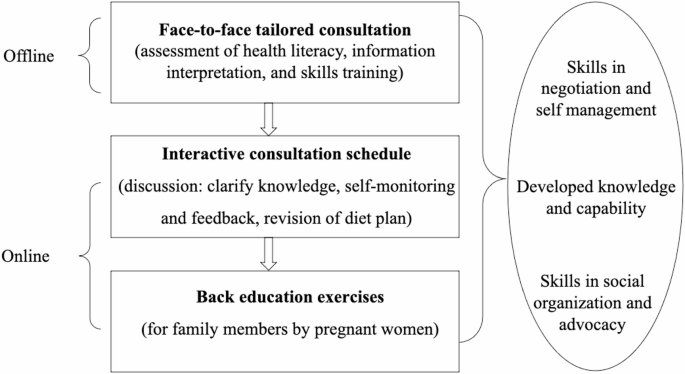
Comprehensive dietary intervention program (CDIP)
The intervention employed a phased approach emphasizing knowledge acquisition, skill development, and practical application, and combined face-to-face consultations with digital support tools. The overall objective was to integrate evidence-based dietary recommendations within the context of participants’ sociocultural environments.
Phase 1: In-person consultation (at 12 weeks of gestation)
A structured 30–40-minute, one-on-one consultation was conducted to establish individualized goals and strategies. Key components included:
Baseline nutritional literacy assessment
Participants’ baseline nutritional literacy was evaluated using a standardized assessment tool. Dimensions included understanding of essential nutrients, self-monitoring capabilities, and decision-making related to dietary choices.
Identification of barriers to information utilization
Participants were encouraged to identify challenges encountered in accessing, interpreting, and applying dietary information. Personalized strategies were introduced to address these barriers, such as the use of simplified educational materials and the engagement of family members.
Guided education on nutritional standards
National dietary standards and recommendations for appropriate GWG were presented in accessible language. Participants were instructed on aligning their dietary choices with these guidelines and were provided culturally relevant examples of nutrient-dense meals.
Skill-Building through practical exercises
Using food models and meal-planning templates, participants practiced translating guidelines into daily meal choices. Strategies to manage common challenges—such as cravings and healthier substitutions—were introduced to facilitate long-term adherence.
Motivational engagement
Participants were informed about subsequent online interventions offering continuous support, flexibility, and prompt feedback. Incentives (e.g., free fetal heart rate monitoring) were provided to foster sustained engagement.
Phase 2: Digital follow-up (16–24 weeks of gestation)
Following the initial consultation, participants transitioned to an online platform designed to reinforce nutritional literacy through interactive content and regular feedback. Key activities included:
Interactive educational modules
Short video segments (10–15 min) addressing self-monitoring of weight gain, macronutrient balance, and energy intake management were made available. The goal was to deepen participants’ comprehension and facilitate practical application of dietary guidelines.
Family engagement and support
An online session at 16 weeks of gestation included participants and primary household meal preparers. This session aimed to improve family-level support for healthy eating practices, ensuring sustained dietary changes within the home environment.
Progress monitoring and feedback
Dietary intake and GWG were monitored biweekly. Personalized feedback was provided through virtual consultations, enabling participants to understand their progress relative to established targets and make informed adjustments.
Adaptive meal planning
Meal plans were regularly updated based on participants’ progress. Nutrient deficiencies, excessive weight gain, or other emerging challenges were addressed by integrating more plant-based options, adjusting portion sizes, or incorporating nutrient-dense foods.
Sustained motivation
Regular reminders, motivational messages, and milestone celebrations were delivered via the online platform to reinforce adherence and acknowledge improvements.
Post-24-week care
Following completion of the intervention at 24 weeks, participants in both the control and intervention arms continued to receive standard antenatal care per national guidelines. Additionally, all participants were offered on-demand dietary consultations until delivery. This service aimed to address residual or newly emerging nutritional concerns, thereby ensuring that maternal and fetal nutritional requirements were met throughout the remainder of the pregnancy.
Treatment fidelity program
Intervention fidelity in this study followed the framework established by the American National Institutes of Health’s Behavior Change Consortium (BCC). The intervention was designed based on a conceptual model of health literacy and relevant behavior change techniques (BCTs). Standardized content covering 11 topics was developed using theoretical evidence, and the frequency and duration of interventions were systematically determined. Midwives delivering the intervention were required to hold registered midwife and maternal-child clinical nursing specialist certificates, undergo training in nutrition and communication skills, and pass qualification assessments. Training was further reinforced through professional retraining by registered dietitians, and obstetrician consultants were assessed on pregnancy weight gain standards. Only those who passed evaluations were permitted to conduct interventions, with weekly monitoring ensuring quality.
The intervention delivery process followed a structured protocol, with providers using a standardized checklist to verify content coverage, appropriate delivery methods, and attendance tracking. Participant adherence was promoted through automated reminders and follow-up calls if needed. After each session, brief interviews assessed participants’ satisfaction, comprehension, and implementation of the intervention. Self-monitoring reports and feedback assisted in evaluating the actual application of intervention strategies. To enhance adherence, participants were incentivized with a free testing program upon completion, and data collection was coordinated with routine antenatal visits to minimize additional hospital visits.
Follow-up and data collection
In this study, the follow-up period was extended to include a third assessment at t3 (before delivery). Data were collected at three time points: baseline (t1) at 12 weeks of gestation before the intervention, post-intervention (t2) at 24 weeks of gestation after the completion of the CDIP, and before delivery (t3) at gestational weeks 37–40. At each time point, nutrition literacy, eating behavior, dietary quality, and GWG were assessed using the validated instruments.
Statistical analysis
The study used SPSS 28.0 for data analysis, applying descriptive statistics (mean, standard deviation, median, and interquartile range) to analyze variable distributions. Baseline data comparisons between groups were conducted using an independent sample t-test for normally distributed continuous variables, while the Mann-Whitney U test or Chi-square test was used for non-normally distributed variables. Within-group comparisons before and after the intervention were analyzed using a paired sample t-test or a paired Wilcoxon signed-rank test. Post-intervention comparisons between groups were conducted using one-way analysis of covariance (One-way ANCOVA) or the Mann-Whitney U test, depending on normality and homogeneity of variance assumptions. Frequency data were analyzed using Fisher’s exact test.
A modified intention-to-treat (mITT) approach was adopted. Participants were analyzed according to their original group allocation, but those with missing outcome data were excluded from the analysis. This approach is acknowledged as a limitation in the discussion.
Human subject protection
The study protocol and informed consent documents were reviewed and approved by REDACTED before the trial commenced. The trial adhered to ethical principles of informed consent, confidentiality, and participant protection. The evaluation researcher provided eligible pregnant women with detailed information on the study’s background, objectives, procedures, potential risks and benefits, alternative treatment options, data confidentiality, compensation, and participant rights. Only after ensuring full comprehension and voluntary agreement did participants sign two identical informed consent forms—one for the researcher and one for themselves.
link





More Stories
Nutrition during pregnancy: Healthy recommendations
Addressing the gaps in nutritional care before and during pregnancy | Proceedings of the Nutrition Society
Sorry, expectant moms: ‘Eating for two’ isn’t really a thing